Category: Pharmaceuticals - Page 4
Switching from Brand to Authorized Generic: Practical Tips for Lower Costs Without Compromising Care
Learn how switching from a brand-name drug to an authorized generic can save you hundreds a year without changing how your medication works. Get practical tips on identifying, verifying, and safely making the switch.
learn moreWhen Your Doctor Might Prescribe Brand-Name Only and Why
Most prescriptions are generics-but sometimes, your doctor prescribes a brand-name drug for a good reason. Learn when it’s medically necessary, why cost shouldn’t be the only factor, and what to ask if you’re unsure.
learn moreMandatory vs Permissive Substitution: How State Laws Control Generic Drug Switching
State laws determine whether pharmacists must or can choose to swap brand-name drugs for generics. These rules affect your costs, safety, and whether you even know you're getting a different pill.
learn moreMethotrexate and NSAIDs: How They Affect Kidney Function and Drug Levels
Methotrexate and NSAIDs can dangerously raise methotrexate levels and harm kidney function. Learn who's at risk, which NSAIDs are safest, and how to prevent life-threatening toxicity.
learn moreOrphan Drug Exclusivity: How Rare-Disease Medicines Get Market Protection
Orphan drug exclusivity gives pharmaceutical companies seven years of market protection for rare-disease treatments, incentivizing development where profits are low. Learn how it works, how it compares to Europe, and why it's both vital and controversial.
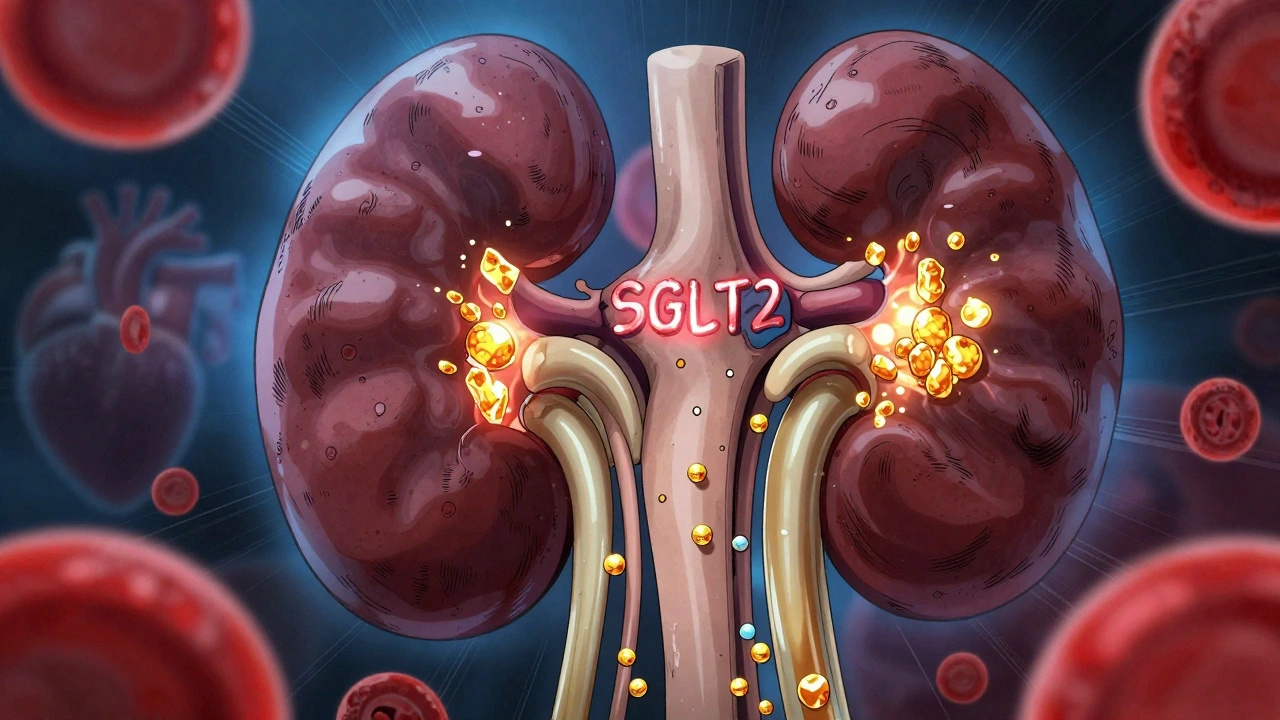
learn moreSGLT2 Inhibitors for Type 2 Diabetes: What You Need to Know About Benefits and Risks
SGLT2 inhibitors like Jardiance and Farxiga offer powerful heart and kidney protection for type 2 diabetes patients-but come with risks like yeast infections, ketoacidosis, and kidney stress. Know who benefits most and what to watch for.
learn moreLitigation in Generic Markets: How Patent Disputes Delay Affordable Medicines
Patent litigation in generic drug markets is delaying affordable medicines, costing billions annually. Learn how the Hatch-Waxman Act, Orange Book listings, and pay-for-delay settlements are shaping access to generics.
learn moreLinezolid and Tyramine: How to Avoid Hypertensive Crisis While Taking This Antibiotic
Linezolid can cause a dangerous blood pressure spike if you eat tyramine-rich foods like aged cheese, cured meats, or red wine. Learn exactly what to avoid, what’s safe, and why you must wait 14 days after treatment.
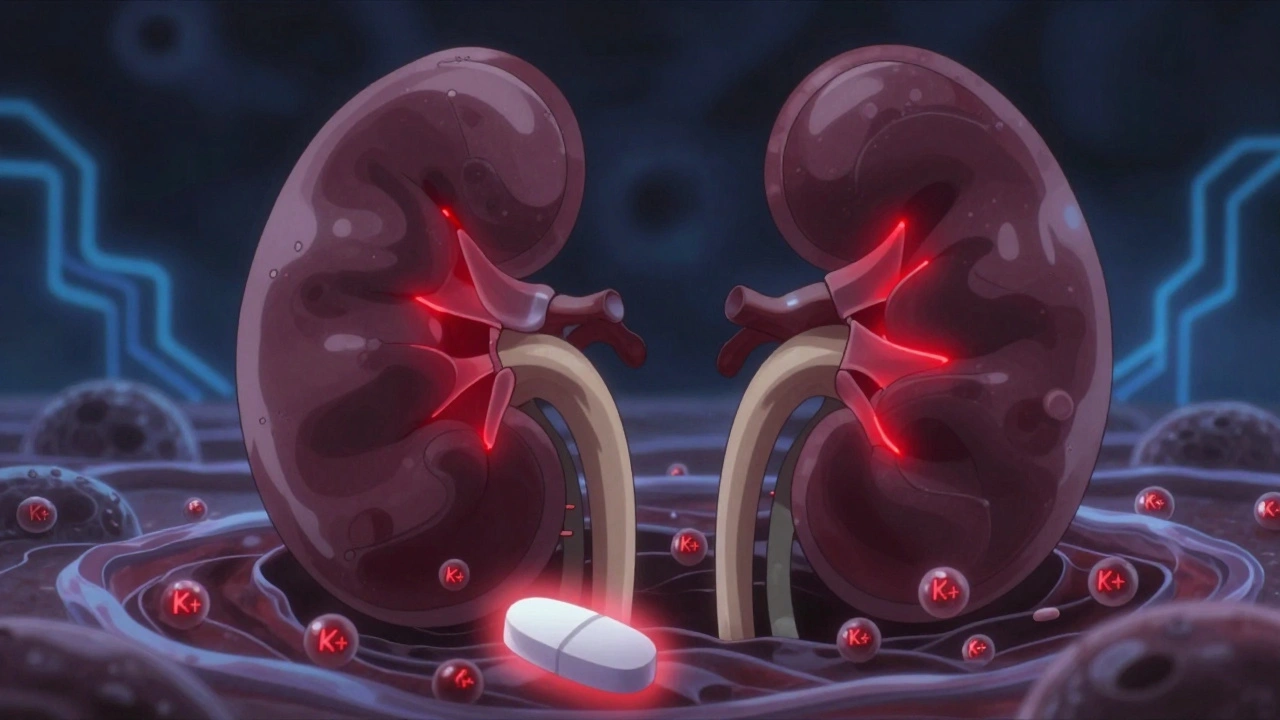
learn moreTrimethoprim and Potassium Levels: How This Common Antibiotic Can Raise Your Risk of Hyperkalemia
Trimethoprim, a common antibiotic, can dangerously raise potassium levels - even in people with healthy kidneys. Learn who’s at risk, how fast it happens, and what safer alternatives exist.
learn moreClinical Outcomes After NTI Generic Switches: What Studies Show
Studies show that switching generics for narrow therapeutic index (NTI) drugs like warfarin, phenytoin, and cyclosporine can affect blood levels and patient outcomes. Learn what the data says-and what you should do.
learn more